State of Agricultural Research on Radioactive Contamination Nuke Info Tokyo No. 159
The Fukushima nuclear disaster caused radioactive substances to be spread widely throughout the environment, and many foods were contaminated with radioactivity. Currently, even though three years have passed since the accident, the mechanisms of transference of radioactivity from soil to plants are not yet well understood. This paper reports on the state of research on radioactive contamination in agriculture, with a focus on radioactive cesium. Note that radioactive iodine-131 has a short half-life (8.02 days), so it is not being detected at this time, and there are few reports of radioactive strontium.*
Tests for Radioactivity in Farm Produce
From April 2012, the allowable concentration of radioactive cesium in foods has been limited to 100 Bq/kg in general foods. The number of investigations into concentrations of radioactive substances conducted nationwide in Japan and the number of cases exceeding the limit are shown in Graph 1.
The number of investigations and the number of cases exceeding the limit fluctuate with the harvesting seasons, rising at times when foods that absorb cesium easily, such as edible wild plants in the spring and mushrooms in the fall, are being harvested. Foods exceeding the limit in 2013 were mushrooms, edible wild plants, soy beans, bamboo shoots, brown rice, buckwheat and komatsuna (Japanese Mustard Spinach).1
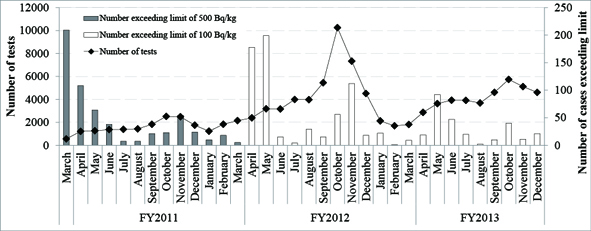 |
| Graph 1. Number of Tests for Radioactive Substances in Foods (farm produce, vegetables) A testing system for rice was instituted in FY2012, when the limit was revised, resulting in a big jump in testing in the fall. (Drawn by CNIC based on results found on the Ministry of Health, Labour and Welfare website.) |
Amounts Transferred from Soil to Plants
There is a transfer coefficient that serves as an indicator of the amount of radioactive substances in soil that will transfer to plants (concentration of radioactive substance in plants ÷ concentration of radioactive substance in soil). Generally, it is high for legumes and root crops, but low for Curcurbitaceae and Brassicaceae.
In FY2011, Fukushima Prefecture found that vegetables with high transfer coefficients when grown outdoors included edamame, with 0.0032 to 0.0040, and sweet potatoes, with 0.0049 to 0.0058. These values indicate that if cultivated in soil with 1,000 Bq/kg, the vegetables produced would contain 3 to 6 Bq/kg. Transfer coefficients for these vegetables were measured again in FY2011 and in FY2012, and the results were reported to have fallen to half what they were.2
The transfere coefficient is thought to be predictable to some degree, depending on the type of plant and soil properties, but there have been cases where it was not.
Occurrence of High Concentrations of Radioactive Cesium in Rice and Investigation into the Causes
–Cannot be Explained by Transfer Coefficients Alone
Graph 2 is a plot of radioactive cesium concentration in brown rice, grown in Fukushima Prefecture in FY2012, versus the concentration in the soil in which it was grown. If in all cases the higher the contamination of the soil, the more cesium was absorbed by brown rice, all of the points on the graph should have been gathered in a line sloping upward toward the right side, but that is not what happened. Even when soil concentrations exceeded 5,000 Bq/kg, the cultivated brown rice was found to contain only a small amount of contamination, while even in fields with soil concentrations of less than 1,000 Bq/kg, highly contaminated brown rice resulted.
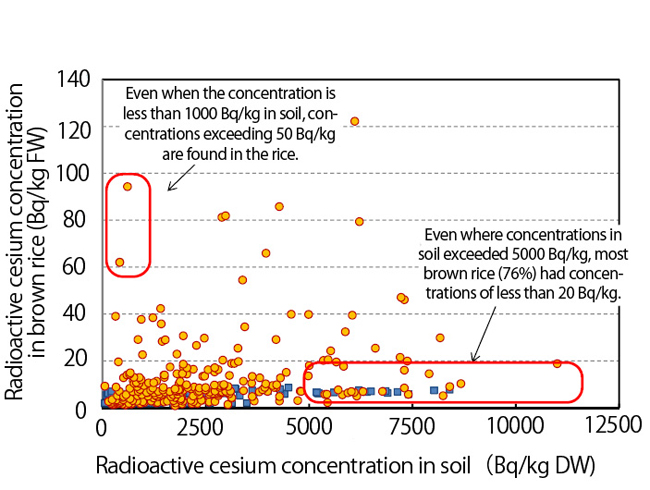 |
| Graph 2. Radioactive cesium concentration in soil versus its concentration in brown rice (Ref. 3). (DW: dry weight, FW: fresh weight) |
–Insufficient Potassium Causes Cesium to be Absorbed
Cesium is chemically similar to potassium, so if a plant has insufficient potassium, it is as if it absorbs cesium by mistake.
Graph 3 shows the relationship between soil potassium concentration and cesium concentration in brown rice, based on a test cultivation of brown rice in FY2012 in an area where brown rice cultivated in FY2011 had been found to have radioactive cesium exceeding 500 Bq/kg. In light of this relationship, it is clear that in soil with plenty of potassium absorption of cesium is impeded, but in soil with insufficient potassium, a high concentration of cesium develops in brown rice.3
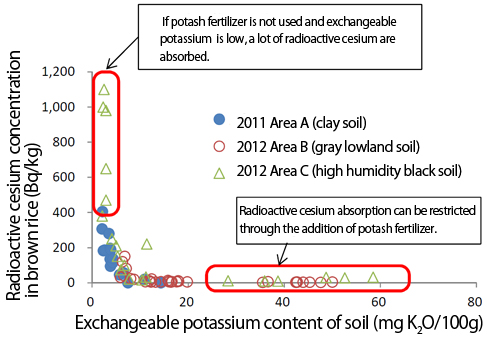 |
| Graph 3. Exchangeable potassium content of soil versus radioactive cesium concentration in brown rice (Ref. 3) |
–Affected by Chemical Form of Cesium
Cesium ions are positively charged, so they are known to adhere easily to the surfaces of minerals or humus from the decomposition of organic matter that is negatively charged. The adhered cesium can be displaced by ammonium or other positive ions, releasing it. Once released, the cesium becomes water-soluble and dissolves. In addition, cesium ions are similar in size to the gaps within molecules of clay minerals, so cesium is known to enter these gaps and get “fixed” to the clay minerals. Once fixed, it is very hard for the cesium to be absorbed by plants.
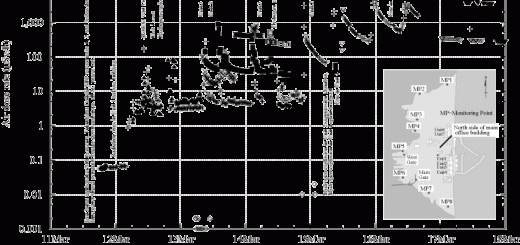
An interesting experimental result was obtained in this regard. When aquatic cultivation of rice was carried out with different concentrations of radioactive cesium, even at a mere 0.1 Bq/liter in water, the cultivated rice leaves had a radioactive cesium concentration of 76 Bq/kg dry weight (Fig. 1). This suggests that the ease of transference to plants is strongly affected by the amount of water-soluble cesium, but not by the total amount of cesium contained in the soil.4
 |
| Fig. 1. Accumulation of radioactive cesium in rice plants grown in water culture medium with different concentrations (Ref. 4). |
–The Paddy Environment and Rice Plant Characteristics
The farming area in Nihonmatsu City in which brown rice was found to have contamination exceeding 500 Bq/kg in September 2011, consisted of valley-bottom paddies surrounded by forest on three sides (with the paddies distributed on terraces along the slopes). The water that the paddies were drawing from the forest contains sufficient potassium and magnesium to grow rice.
A survey confirmed that the soil potassium concentration in the paddies was low, and the clay content of the soil was small.
Although the paddies were receiving water with abundant nutrients, for some reason the soil did not contain sufficient potassium to prevent cesium absorption. Even in areas where the soil of valley-bottom paddies originally contained too few nutrients, there were agricultural techniques that made rice cultivation possible using water from forests. It is said that the valley-bottom paddy farmers of Nihonmatsu City normally did not use very much potassium fertilizer.5,6 Therefore, the soil had perhaps been chronically lacking in potassium.
The rice plants themselves were discovered to have differing characteristics. For the most part, the cesium concentration in rice leaves was higher in the lower leaves and lower in the uppermost leaves. In cases in which the brown rice had high concentrations of cesium, however, the upper leaves of the plants had high concentrations. The upper leaves grow during summer, so perhaps there was a particular source of cesium for some reason in those paddies in the summer. After that, the ears must have formed with high concentrations of cesium.
The causes are currently being actively studied, with possibilities noted such as hot temperatures in summer promoting decomposition of organic matter, including fallen leaves containing radioactive cesium, and water carrying that cesium in from surrounding areas.
Airborne Transport of Chemical Compounds of Radioactive Cesium
The chemical form of the radioactive cesium that was carried by winds from the Fukushima nuclear power plant is not well understood. Research has been done analyzing fine atmospheric dust (aerosols) collected in Tsukuba City after the accident. Some of the aerosol particles had diameters of several micrometers, and others, 0.5 to 0.7 micrometers. An elemental analysis detected cesium and sulfate ions together with the latter, smaller aerosol particles. Perhaps the cesium was falling to the ground as a sulfate salt.6 On the other hand, high concentrations of radioactive cesium have been detected in spherically shaped particles of several micrometers in diameter in some areas near the Fukushima NPP.
After the accident, many kinds of vegetables were found with iodine, cesium and other radioactive substances sticking to their surfaces and their shipments were halted. Investigations on methods to remove the radioactive substances from the surfaces revealed that about 60% could be eliminated by washing with water, but that the addition of physical stimuli such as ultrasonic cleaning did not change this ratio. The use of acid, alkali or alcohol failed to produce a high removal ratio, but the use of a reducing agent (1% sodium thiosulfate) used as an antioxidant for foods increased the elimination of iodine. This was observed to be due to conversion of the hard-to-dissolve iodine molecules (I2) to water-soluble iodine ions (I–).7
Investigation of Circular Agriculture
As an example of circular agriculture, hay and other feed is grown in soil; livestock eat that and produce manure; the manure, together with plant and other waste, is used to produce compost; and the compost is added to the soil to provide nutrients for plants, as illustrated by the relationships in Fig. 2. After the nuclear accident, the allowable amount of radioactive cesium in fertilizers was limited to 400 Bq/kg, and in feed for cattle, hogs and other livestock, to 300 Bq/kg, and in some cases, the circular relationship was broken.
In FY2011, the muscles of goats continuously given feed with 3,900 Bq/kg were found to contain cesium of 130 Bq/kg, and their manure contained 150 Bq/kg, resulting in compost containing 890 Bq/kg.
Research findings on plant cultivation using contaminated compost show that cultivation of vegetables solely with compost of about 800 Bq/kg produce eggplant, maize, soybeans and ginger with less radioactive cesium than the detection limit of 20 Bq/kg. In order to produce the highest degree of contamination, the compost was not mixed with soil, but used alone for cultivation.8 It bears noting that if the compost used is not fully matured, it may contain ammonium ions, which displaces and frees cesium adhering to the soil, possibly increasing the amount of cesium that can be taken up easily by plants, so caution is necessary if using compost that is not fully matured.
 |
| Fig. 2. Conceptual diagram of sustainable agriculture (example). |
Efforts to Curb Exposure During Farm Work
If tilling is not carried out, almost all of the radioactive cesium is fixed within the top 5 cm from the soil surface.6 In this case, if the surface layer of the soil is removed, the air dose rate decreases, but this requires removal of fertile soil, and it also creates problems on where to put the discarded radioactive waste.
In Fukushima Prefecture, efforts have been made to decrease the amount of radioactive cesium in the surface layer by deep tilling of the soil or by interchanging the upper and lower soil layers. When “plow tilling” was tried, in which the radioactive cesium in the surface layer was plowed into the lower layer, the radiation levels in flooded paddies were reduced by about 50%, and the greatest effects were achieved in fields, where reductions of nearly 90% were reported (Table 1).
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Table 1. Plow tilling air dose rates and their reduction (at 100 cm above ground surface).(Ref. 2) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
The natural world is subject to complex influences. Observing nature in detail, following up on slight hints, setting up experiments, and elucidating the mechanisms of radioactive contamination require serious work. In practical field tests, long periods of time are needed between planting seeds and harvesting crops, and in the case of rice, no more than one verification can be accomplished each year. It is thought that a certain amount of time will be needed in order to ascertain the mechanisms of the transference of radioactive substances into food crops. Also, reducing exposures during farm work is considered an issue of importance to the continuation of agriculture.
(Nobuko Tanimura, CNIC)
[note]
*When distributed foods were investigated in Fukushima, Iwate, Tochigi, Niigata, Ibaraki, Kanagawa, Saitama and Kochi prefectures, radioactive strontium was detected in seven of the 20 foods tested. The concentrations ranged from 0.016 to 0.039 becquerels per kilogram (Bq/kg), which amounted to about 1% of the radioactive cesium concentration.
“Results of testing for radioactive strontium and plutonium in foods (results from Feb-May, 2012)” found on the Japan Ministry of Health, Labour and Welfare website (www.mhlw.go.jp/stf/houdou/0000028846.html)– in Japanese
[References]
(1) “Coping with radioactive substances in food” (Shokuhinchu no hoshaseibusshitsu e no taio). Japanese Ministry of Health, Labour and Welfare website. www.mhlw.go.jp/shinsai_jouhou/shokuhin.html
(2) “Regarding research on radioactive substances in the field of agriculture” (Nogyobunya ni okeru hoshaseibusshitsu shiken kenkyu ni tsuite). Fukushima Agricultural Technology Centre. www4.pref.fukushima.jp/nougyou-centre/kenkyuseika/kenkyu_seika_radiologic.html
(3) “Regarding causes of and countermeasures to high concentrations of radioactive cesium in rice” (Hoshasei seshium nado no takai kome ga hassei suru yoin to sono taisaku ni tsuite). Japanese Ministry of Agriculture, Forestry and Fisheries. www.maff.go.jp/j/kanbo/joho/saigai/s_seisan_1.html
(4) Nemoto Keisuke. “New knowledge on absorption by crops” (Sakumotsu no shinchiken). Gakujutsu no Doko, Oct. 2012, pp.22-26.
(5) “Results of an investigation of paddies in Obama-cho, Nihonmatsu City, on Oct. 17, 2011” (Nihonmatsu-shi Obama-cho no suiden ni okeru chosa kekka Heisei 23 nen 10 gatsu 17 nichi). Agriculture, Forestry and Fisheries Industries of Fukushima Prefecture. www.pref.fukushima.jp/keieishien/kenkyuukaihatu/gijyutsufukyuu/05gensiryoku/231017_obama.pdf
(6) Nakanishi Yuko (2013), Dojo Osen Fukushima no Hoshaseibusshitsu no Yukue (Soil Contamination: The whereabouts of Fukushima’s radioactive substances), NHK Books, 220 pp.
(7) Vegetable Analysis Group, Ad Hoc Committee on Safety Measures for Radioactive Iodine and Cesium, Japanese Society of Radiation Safety Management. “Interim report on methods of removing radioactive substances adhering to vegetables contaminated by fallout from the Fukushima Daiichi nuclear accident” (Fukushima Daiichi genpatsujiko ni yotte osen sareta yasai ni fuchaku shita hoshaseibusshitsu no jokyoho ni kansuru chukan hokoku). Isotope News. 2011, No. 689, pp. 55-58.
(8) Report by Manabe Noboru, et al., at a study briefing session on the effects of radioactivity on agriculture, livestock and marine products, The University of Tokyo School of Agriculture and Life Sciences.